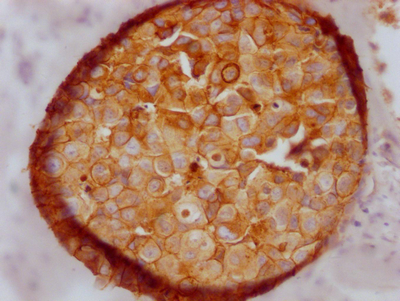
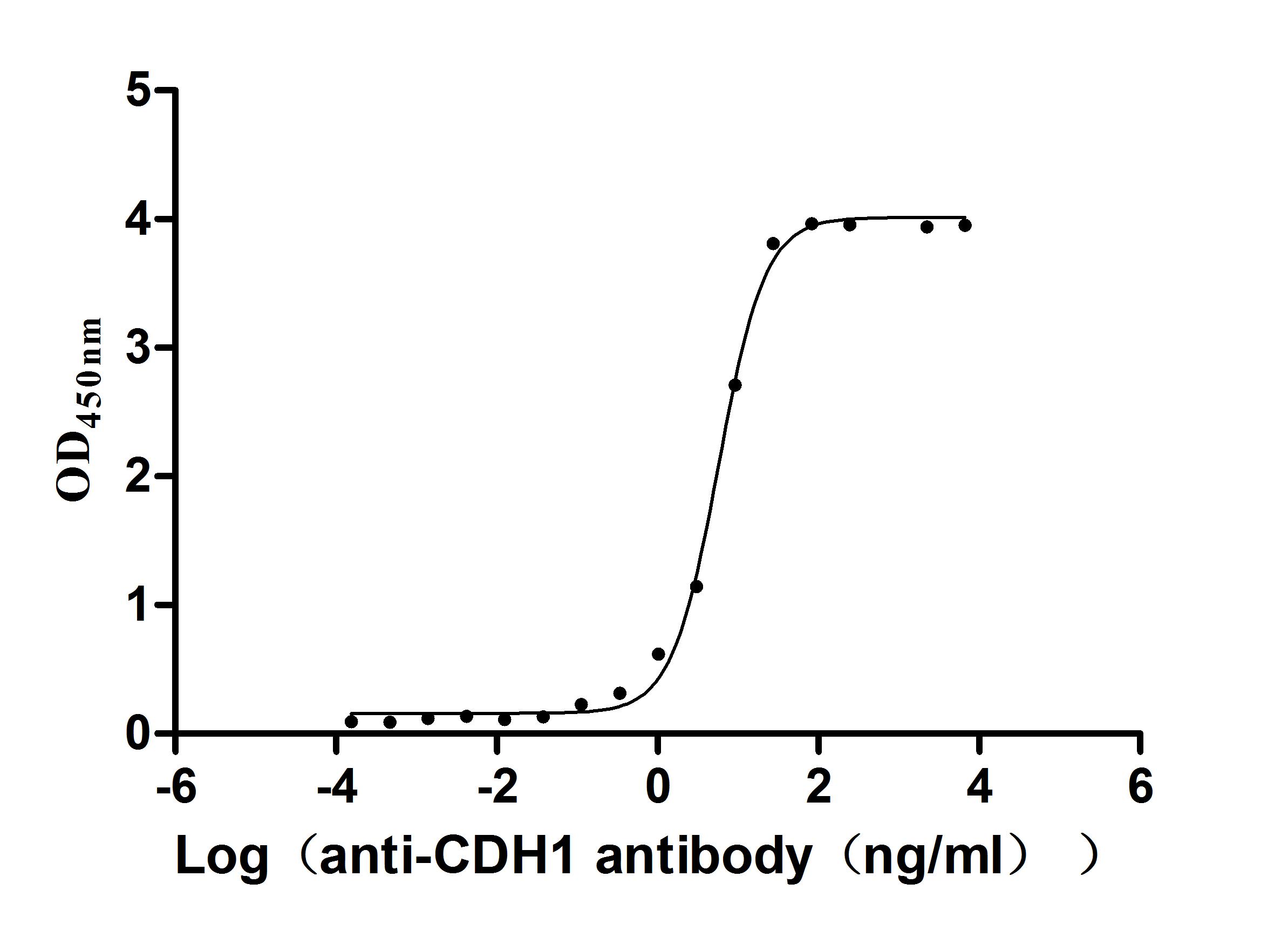
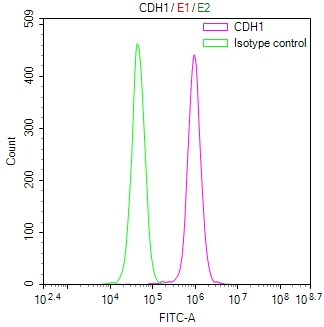
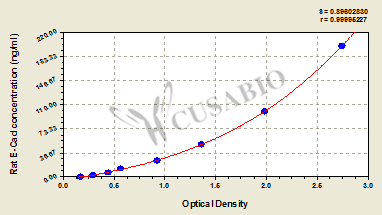
CDH1/E-cadherin:從黏附調控到腫瘤靶向治療的核心靶點研究進展
日期:2026-03-17 08:52:24
E-cadherin由CDH1基因編碼,是上皮細胞鈣依賴性黏附分子,在維持上皮結構完整性、細胞間通訊與組織穩態中處于核心地位 [1,2]。CDH1功能受損常導致細胞間黏附松解并觸發上皮-間充質轉化(EMT)等關鍵表型變化,是多種腫瘤發生發展的重要驅動因素 [3]。本文圍繞CDH1的結構與生理功能、多層次調控、關鍵通路、疾病譜和靶向藥物最新研究進展進行結構化梳理,以期為您的研究帶來幫助。
1. 背景與研究意義:CDH1為何是“黏附—信號—疾病”樞紐
2. CDH1/E-cadherin的結構與細胞-細胞黏附:從分子裝配到力學耦聯
3. 多層次調控機制:決定E-cadherin“表達水平與功能狀態”的關鍵環節
1. 背景與研究意義:CDH1為何是“黏附—信號—疾病”樞紐
E-cadherin通過形成黏附連接(adherens junctions)將相鄰細胞機械耦聯,并與肌動蛋白骨架連接,參與細胞遷移、分化與信號轉導 [2]。因此,E-cadherin通常被視為重要腫瘤抑制因子,其缺失可削弱細胞黏附并促進侵襲轉移 [3]。除宏觀組織結構外,E-cadherin還參與更精細的空間組織:在角質形成細胞中,E-cadherin缺失可造成微尺度細胞分離(micro-demixing),提示黏附在微觀組織均質性/模式形成中具有“精細調參”作用 [4]。
臨床上,CDH1致病性生殖系突變與遺傳性彌漫性胃癌(HDGC)高度相關,攜帶者具有顯著升高的彌漫性胃癌終生風險 [5-9],并且在特定人群(如新西蘭毛利人)中被證實是早發彌漫性胃癌高發的重要遺傳因素 [5]。除編碼區突變外,下游調控序列缺失(如CDH1-TANGO6缺失)也可顯著下調CDH1表達并導致極早發、高外顯率的彌漫性胃癌 [10];在散發性胃癌中,CDH1遺傳變異與表觀遺傳改變(如啟動子甲基化)同樣與致癌過程密切相關 [11]。此外,彌漫性胃癌中細胞-細胞/細胞-基質黏附依賴性的“逃逸”與RHO信號擾動(如RHOA突變或ARHGAP融合)相關,強調黏附改變與細胞內信號重塑存在協同關系 [12]。
值得注意的是,CDH1在不同腫瘤背景下可能呈現差異性作用。例如在乳腺癌中,CDH1可出現表達上調,并與分期、轉移、干細胞特性及不良預后相關,提示其在特定背景下可能呈現促腫瘤效應 [13];而在治療選擇有限的三陰性乳腺癌中,CDH1缺陷較為常見,推動了“CDH1缺陷型腫瘤”靶向策略的探索 [14]。
2. CDH1/E-cadherin的結構與細胞-細胞黏附:從分子裝配到力學耦聯
E-cadherin是跨膜糖蛋白,胞外區由多個cadherin repeats構成,在鈣離子存在下穩定并介導同型結合,是黏附連接形成的基礎 [1]。其胞內區與β-catenin等銜接蛋白結合并連接肌動蛋白骨架,從而實現力學傳遞與組織尺度的機械耦聯 [17,2]。相關生物物理模型將黏附復合物視作可傳遞阻力/張力的“彈簧樣結構”,能夠解釋細胞極化、振蕩動力學以及超細胞應力鏈等多細胞行為,提示“黏附—骨架”并非靜態連接,而是雙向耦合的動力系統 [2]。
結構層面的致病變異可直接破壞黏附功能。例如G212E錯義突變會顯著影響E-cadherin穩定性、定位與黏附能力,導致組織結構紊亂并削弱抗侵襲性 [19]。此外,E-cadherin功能也受復雜信號網絡調控:如PAK5可參與維持細胞-細胞黏附完整性,提示黏附復合物并非“結構件”,而是受激酶網絡持續調節的功能模塊 [20]。
3. 多層次調控機制:決定E-cadherin“表達水平與功能狀態”的關鍵環節
3.1 遺傳與表觀遺傳調控
在HDGC及散發性胃癌中,CDH1可因編碼區突變、調控序列缺失或啟動子甲基化等機制導致表達下降或功能缺失 [5,10,11]。在炎癥背景下,CDH1位點CpG甲基化增加也被報道與黏膜炎癥相關聯,提示慢性炎癥可能通過表觀遺傳途徑削弱上皮屏障 [24]。此外,乳腺癌中也存在CDH1異常甲基化與表達缺失的證據 [25]。
3.2 轉錄后調控與非編碼RNA網絡
多種miRNA/lncRNA被用于解釋CDH1在不同腫瘤中的動態變化。例如,miR-92a-3p在膠質瘤及膠質瘤干樣細胞中可通過靶向CDH1/β-catenin并影響Notch-1/Akt信號,參與腫瘤表型調控 [26];lncRNA SNHG1與hnRNPL形成復合體并共同調控CDH1,從而促進前列腺癌生長與轉移 [32]。
3.3 翻譯后修飾與蛋白互作
糖基化是影響E-cadherin活性與細胞行為的重要機制。在胰腺癌細胞中,ST3Gal III改變E-cadherin唾液酸化模式并降低細胞-細胞聚集能力,同時增強侵襲遷移相關信號(如FAK Tyr397磷酸化),提示“糖鏈-黏附-遷移”之間存在可觀測的功能鏈條 [22]。互作層面,MCC蛋白可與E-cadherin及β-catenin互作并增強結直腸癌細胞黏附,提示黏附復合體穩定性還依賴腫瘤抑制網絡的協同 [21]。
4. 關鍵信號通路與細胞表型:從EMT到微環境適應
CDH1缺陷最典型的后果是EMT相關表型增強。已有研究提出,SPHK1可通過促進自噬-溶酶體途徑降解CDH1/E-cadherin從而誘導EMT,提示“代謝酶—自噬—黏附降解”的串聯機制可能參與肝癌進展 [31]。另一方面,CDH1表達也與代謝重編程存在聯系:E-cadherin可誘導絲氨酸合成以支持乳腺癌進展和轉移,提示其在特定背景下可能通過代謝路徑促進腫瘤適應 [28]。ZHX2缺陷可富集雜合型MET細胞并通過調控E-cadherin表達影響EMT/MET動態平衡,強調CDH1并非簡單“開/關”,而可能參與多狀態轉換 [33]。
在通路層面,CDH1常與Wnt/β-catenin、PI3K/AKT/mTOR、MAPK/ERK及TGF-β/Smad等網絡交織。既往研究在食管癌中討論了Wnt/β-catenin與TGF-β-Smad通路的表觀遺傳失調及其對預后的影響 [3];在結直腸癌中,WNT通路組分也存在遺傳與表觀遺傳改變并與微衛星不穩定性分層相關 [30]。
此外,CDH1異常還與上皮屏障破壞相關:在SARS-CoV-2感染的Caco-2腸上皮模型中,CDH1/E-cadherin表達與可溶性E-cadherin釋放受到影響,并被討論為腸道表現相關的潛在生理病理基礎之一 [29]。
5. 相關疾病:從高外顯率遺傳綜合征到多癌種與炎癥/感染上皮病理
5.1 遺傳性彌漫性胃癌(HDGC):以CDH1缺陷為核心的高風險疾病譜
HDGC是CDH1研究中最具“遺傳—機制—臨床管理”閉環特征的場景。現有研究強調:在新西蘭毛利人群中,生殖系CDH1突變被證實顯著貢獻于早發彌漫性胃癌的高發生頻率,提示其在特定遺傳背景/人群結構下具有重要公共衛生意義 [5]。除經典編碼區突變外,CDH1及其下游調控序列的聯合缺失(CDH1-TANGO6缺失)可造成CDH1表達顯著下降,并與極早發且高外顯率彌漫性胃癌相關,提示“編碼區之外的調控區域”同樣可能決定疾病負擔 [10]。
在散發性胃癌中,CDH1相關的遺傳變異與表觀遺傳改變(例如調控區改變與啟動子甲基化)被一并討論為影響CDH1表達與致癌過程的重要因素,提示HDGC與散發性胃癌之間并非完全割裂,而可能在機制上存在“同軸不同強度”的連續譜 [11]。基于胃癌類器官模型的研究強調,疾病進展過程中可出現對細胞-細胞與細胞-基質黏附依賴性的“逃逸”,并伴隨RHO信號擾動(如RHOA突變或ARHGAP融合)[12]。這類證據把“CDH1缺陷導致黏附失衡”與“細胞內信號重塑”連接起來,為理解彌漫性胃癌的侵襲性生物學行為提供機制線索 [12]。
5.2 乳腺癌:CDH1既可能是缺失驅動,也可能呈現情景依賴的“反直覺表達”
乳腺癌中CDH1呈現更強的異質性,CDH1生殖系突變與遺傳性小葉型乳腺癌相關,被用于討論遺傳性腫瘤風險譜與遺傳咨詢路徑 [9]。此外,家族性乳腺癌風險也被報道與CDH1等位基因SNP相關性有關 [27]。在部分研究中,CDH1可能出現表達上調,并與分期、轉移、干細胞特性及不良預后相關,提示其在某些背景下可能呈現促腫瘤效應 [13]。這意味著在乳腺癌語境下,不能簡單用“E-cadherin高=抑癌、低=促癌”概括,其臨床解釋往往需要結合腫瘤分型與分子網絡 [13]。在三陰性乳腺癌等治療選擇有限場景中,CDH1缺陷更容易被納入“可干預脆弱性”的討論,并推動聯合抑制策略探索 [14]。
5.3 結直腸癌:易感基因證據、黏附復合體穩態與炎癥相關腫瘤免疫
結直腸癌相關證據鏈條更偏向“遺傳易感—網絡穩態—腫瘤免疫背景”,GWAS研究把CDH1納入結直腸癌遺傳易感基因圖譜,提示其在群體層面具有風險相關性 [15]。MCC蛋白與E-cadherin/β-catenin互作可增強結直腸癌細胞黏附,強調CDH1相關表型不僅由單基因決定,也與黏附復合體伙伴蛋白網絡有關 [21]。在炎癥性腸病相關結直腸癌中,IBD相關基因被用于預后與腫瘤免疫含義分析,CDH1亦被納入相關討論框架 [16]。與此相呼應,結直腸癌中的WNT通路組分改變及其與微衛星不穩定性分層的關系,為解釋上皮穩定性破壞與信號重編程提供了宏觀通路層面的背景 [30]。
5.4 食管癌與癌前病變:CDH1/CTNNB1表達下降與轉移、預后相關
在食管癌中,CDH1或CTNNB1表達降低與淋巴結轉移及不良預后相關,這一觀察把“黏附復合體失衡”與更不利的臨床結局聯系起來 [17]。同時,關于Wnt/β-catenin與TGF-β-Smad通路的表觀遺傳失調被用于解釋食管癌的預后差異,提示CDH1改變往往與更大范圍的通路層級異常并行出現 [3]。
在癌前病變層面,口腔扁平苔蘚中EMT相關蛋白(含E-cadherin)的表達改變提示細胞連接紊亂可能與病變演進相關,為“黏附改變的早期提示意義”提供了證據線索 [18]。
5.5 炎癥與感染相關上皮屏障:甲基化改變與可溶性E-cadherin釋放
CDH1不僅與腫瘤相關,也與上皮屏障狀態密切相關:在炎癥背景下,CDH1位點CpG甲基化增加與黏膜炎癥相關聯,提示慢性炎癥可能通過表觀遺傳途徑影響上皮黏附與屏障穩態 [24]。在SARS-CoV-2感染的Caco-2腸上皮模型中,CDH1/E-cadherin表達及可溶性E-cadherin釋放發生變化,被用于討論腸道表現相關的病理基礎之一 [29]。與檢測相關的證據還包括:尿液中可溶性E-cadherin片段升高被認為可能反映上皮腫瘤細胞的剪切/脫落過程,為非侵入性標志物提供線索 [23]。
6. CDH1靶向藥物最新研究進展
目前靶向CDH1的藥物研發主要處于臨床前及早期發現階段,涵蓋小分子化藥、生物藥、外泌體等多種類型。主要探索方向包括腦惡性膠質瘤、乳腺癌及多囊疾病等,涉及沈陽藥科大學、哈佛大學、四川省腫瘤醫院等多家機構。
| 藥物 | 類型 | 適應癥 | 研發階段 | 研發機構 |
|---|---|---|---|---|
| AL-GDa62 | 小分子抑制劑 | CDH1缺陷型胃癌 | 臨床前 | 新西蘭奧塔哥大學 |
| Dasatinib | 多激酶抑制劑 | CDH1缺陷型腫瘤 | 臨床前 | 多個研究機構 |
| FAK抑制劑 + ROS1抑制劑 | 聯合療法 | CDH1缺陷型癌癥 | 臨床前 | 多個研究機構 |
| 外泌體遞送CDH1 mRNA | 基因治療 | 上皮屏障損傷修復 | 探索階段 | 國內科研機構 |
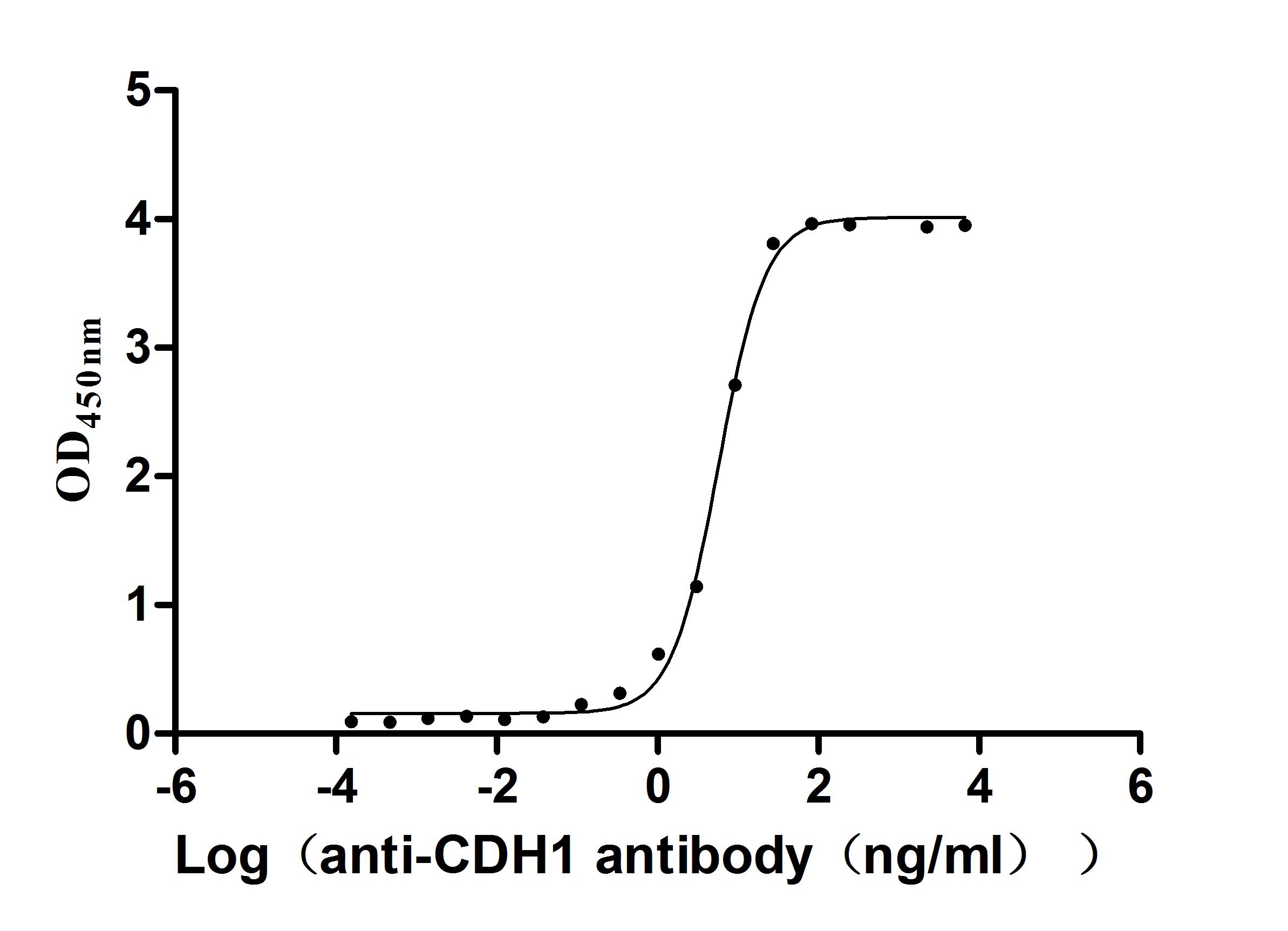
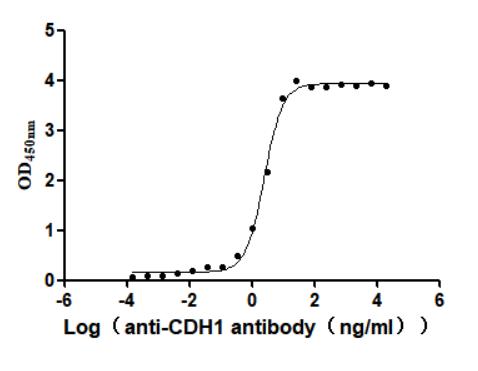
7. CDH1研究工具推薦:重組蛋白、抗體與ELISA試劑盒選型指南
華美生物提供CDH1重組蛋白、抗體及ELISA試劑盒產品,助力您進行相關機制研究及靶向藥物開發。
[1] Anna Zhigun, Mabel Lizzy Rajendran.(2023). Modelling non-local cell-cell adhesion: a multiscale approach.
[2] XinXin Du, Ido Lavi, Michael J. Shelley.(2025). Collective multicellular patterns arising from cadherin-linked cytoskeletal domains.
[3] Virendra Singh, A. Singh, I. Sharma, L. Singh, J. Sharma, B. B. Borthakar, A. Rai, A. Kataki, S. Kapur, S. Saxena.(2019). Epigenetic deregulations of Wnt/β-catenin and transforming growth factor beta-Smad pathways in esophageal cancer: Outcome of DNA methylation.
[4] Preeti Sahu, Daniel M. Sussman, Matthias Rubsam, Aaron F. Mertz, Valerie Horsley, Eric R. Dufresne, Carien M. Niessen, M. Cristina Marchetti, M. Lisa Manning, J. M. Schwarz.(2019). Small-scale demixing in confluent biological tissues.
[5] C. Hakkaart, L. Ellison-Loschmann, R. Day, A. Sporle, Jonathan Koea, Pauline Harawira, Soo Cheng, M. Gray, Tracey Whaanga, N. Pearce, P. Guilford.(2018). Germline CDH1 mutations are a significant contributor to the high frequency of early-onset diffuse gastric cancer cases in New Zealand Māori.
[6] I. Chen, Lesley Mathews-Greiner, Dandan Li, A. Abisoye-Ogunniyan, Satyajit Ray, Yansong Bian, V. Shukla, Xiaohu Zhang, Rajarashi Guha, Craig J. Thomas, B. Gryder, A. Zacharia, J. Beane, S. Ravichandran, M. Ferrer, U. Rudloff.(2017). Transcriptomic profiling and quantitative high-throughput (qHTS) drug screening of CDH1 deficient hereditary diffuse gastric cancer (HDGC) cells identify treatment leads for familial gastric cancer.
[7] Nicola Bougen-Zhukov, Lyvianne Decourtye-Espiard, Wilson Mitchell, Kieran Redpath, J. Perkinson, T. Godwin, M. Black, P. Guilford.(2022). E-Cadherin-Deficient Cells Are Sensitive to the Multikinase Inhibitor Dasatinib.
[8] T. Godwin, S. Kelly, Tom Brew, Nicola Bougen-Zhukov, A. Single, Augustine Chen, Cassie E Stylianou, L. Harris, Sophie K Currie, Bryony J. Telford, Henry Beetham, G. Evans, M. Black, P. Guilford.(2018). E-cadherin-deficient cells have synthetic lethal vulnerabilities in plasma membrane organisation, dynamics and function.
[9] G. Corso, M. Intra, C. Trentin, P. Veronesi, V. Galimberti.(2016). CDH1 germline mutations and hereditary lobular breast cancer.
[10] C. São José, J. Garcia-Pelaez, Marta Ferreira, O. Arrieta, Ana André, Nelson Martins, Samantha Solís, B. Martínez-Benítez, M. Ordoñez-Sánchez, M. Rodríguez-Torres, Anna K. Sommer, Iris B A W Te Paske, C. Caldas, M. Tischkowitz, María Teresa Tusié, Stefan Gabriel Sérgio Richarda M. Gareth Susana José Luzi Aretz Capella Castedo de Voer Evans Fernandes Garc, S. Aretz, G. Capellá, S. Castedo, Richarda M. de Voer, Gareth Evans, Susana Fernandes, J. Garcia-Pelaez, L. Garrido, E. Holinski-Feder, N. Hoogerbrugge, D. Huntsman, A. Jahn, C. Kets, A. Laner, M. Ligtenberg, Andrea Meinhardt, A. Mensenkamp, Carla Oliveira, S. Peters, I. Quintana, E. Schröck, Anna K. Sommer, I. Spier, L. Spruijt, V. Steinke-Lange, I. T. Paske, M. Tischkowitz, L. Valle, R. S. van der Post, Y. V. van Herwaarden, W. V. van Zelst-Stams, D. William, N. Hoogerbrugge, German Demidov, Richarda M. de Voer, S. Laurie, Carla Oliveira.(2023). Combined loss of CDH1 and downstream regulatory sequences drive early-onset diffuse gastric cancer and increase penetrance of hereditary diffuse gastric cancer.
[11] G. Tedaldi, C. Molinari, C. São José, Rita Barbosa-Matos, A. André, R. Danesi, V. Arcangeli, M. Ravegnani, L. Saragoni, P. Morgagni, F. Rebuzzi, M. Canale, S. Pignatta, Elisa Ferracci, G. Martinelli, G. Ranzani, Carla Oliveira, D. Calistri, P. Ulivi.(2021). Genetic and Epigenetic Alterations of CDH1 Regulatory Regions in Hereditary and Sporadic Gastric Cancer.
[12] Yin Tong, P. Cheng, Chung Sze Or, Sarah S K Yue, H. Siu, S. Ho, S. Law, W. Tsui, D. Chan, Stephanie Ma, Siu Po Lee, Annie S. Y. Chan, April S Chan, Shui Wa Yun, Ho Sang Hui, S. Yuen, S. Leung, Helen H. N. Yan.(2022). Escape from cell-cell and cell-matrix adhesion dependence underscores disease progression in gastric cancer organoid models.
[13] D. Xie, Yiyu Chen, Xue Wan, Jingyuan Li, Qin Pei, Yanan Luo, Jinbo Liu, Ting Ye.(2022). The Potential Role of CDH1 as an Oncogene Combined With Related miRNAs and Their Diagnostic Value in Breast Cancer.
[14] Jiaming Gao, Yunying Yao, Zaiqi Wang, Baoyuan Zhang, R. Ren.(2023). Abstract 5821: Synergism of FAK and ROS1 inhibitors in the treatment of CDH1-deficient cancers mediated by FAK-YAP-TRX signaling.
[15] Da-Tian Bau, Ting-Yuan Liu, Jai-Sing Yang, William Tzu-Liang Chen, Chia‐Wen Tsai, Wen‐Shin Chang, Tao-Wei Ke, Chi-Chou Liao, Yu-Chia Chen, Yen-Ting Chang, Fuu-Jen Tsai.(2024). Characterizing Genetic Susceptibility to Colorectal Cancer in Taiwan Through Genome‐Wide Association Study.
[16] Di Wang, Biao Xie.(2022). Prognostic and tumor immunity implication of inflammatory bowel disease-associated genes in colorectal cancer.
[17] H. Ishiguro, T. Wakasugi, Yukio Terashita, N. Sakamoto, Tatsuya Tanaka, K. Mizoguchi, H. Sagawa, T. Okubo, H. Takeyama.(2016). Decreased expression of CDH1 or CTNNB1 affects poor prognosis of patients with esophageal cancer.
[18] L. Hämäläinen, Y. Soini, S. Pasonen-Seppänen, M. Siponen.(2019). Alterations in the expression of EMT-related proteins claudin-1, -4 and -7, E-cadherin, TWIST1 and ZEB1 in oral lichen planus..
[19] J. Figueiredo, F. Mercadillo, Soraia Melo, A. Barroso, Margarida Gonçalves, J. Diaz-Tasende, Patrícia Carneiro, L. Robles, F. Colina, C. Ibarrola, J. Perea, Eurico Morais-de-Sá, R. Seruca, M. Urioste.(2021). Germline CDH1 G212E Missense Variant: Combining Clinical, In Vitro and In Vivo Strategies to Unravel Disease Burden.
[20] A. F. Ismail, Sevil Oskay Halacli, N. Babteen, M. De Piano, T. Martin, W. Jiang, Muhammad S. Khan, P. Dasgupta, C. Wells.(2017). PAK5 mediates cell: cell adhesion integrity via interaction with E-cadherin in bladder cancer cells..
[21] F. Benthani, F. Benthani, D. Herrmann, D. Herrmann, P. N. Tran, P. N. Tran, L. Pangon, L. Pangon, M. Lucas, A. Allam, N. Currey, S. Al‐Sohaily, Marc Giry-Laterrière, J. Warusavitarne, P. Timpson, P. Timpson, M. R. Kohonen-Corish, M. R. Kohonen-Corish, M. R. Kohonen-Corish.(2018). ‘MCC’ protein interacts with E-cadherin and β-catenin strengthening cell–cell adhesion of HCT116 colon cancer cells.
[22] Sònia Bassagañas, S. Carvalho, A. M. Dias, Marta Pérez-Garay, M. R. Ortiz, J. Figueras, C. Reis, S. Pinho, R. Peracaula.(2014). Pancreatic Cancer Cell Glycosylation Regulates Cell Adhesion and Invasion through the Modulation of α2β1 Integrin and E-Cadherin Function.
[23] M. Katayama, S. Hirai, M. Yasumoto, K. Nishikawa, S. Nagata, M. Otsuka, K. Kamihagi, I. Kato.(1994). Soluble fragments of e-cadherin cell-adhesion molecule increase in urinary-excretion of cancer-patients, potentially indicating its shedding from epithelial tumor-cells..
[24] Charles de Ponthaud, Solafah Abdalla, M. Belot, Xiaojian Shao, Christophe Penna, A. Brouquet, Pierre Bougnères.(2024). Increased CpG methylation at the CDH1 locus in inflamed ileal mucosa of patients with Crohn disease.
[25] Asia Asiaf, S. T. Ahmad, S. Aziz, A. Malik, Zubaida Rasool, A. Masood, M. Zargar.(2014). Loss of expression and aberrant methylation of the CDH1 (E-cadherin) gene in breast cancer patients from Kashmir..
[26] Hang Song, Yao-quan Zhang, Na Liu, Sheng Zhao, Y. Kong, Liudi Yuan.(2016). miR-92a-3p Exerts Various Effects in Glioma and Glioma Stem-Like Cells Specifically Targeting CDH1/β-Catenin and Notch-1/Akt Signaling Pathways.
[27] M. Bagherpour, Kamelia Gharibzad, H. Rassi.(2018). Association of CDH1 and TERT Single-Nucleotide Polymorphisms with Susceptibility to Familial Breast Cancer Risk..
[28] Geonhui Lee, Claudia Wong, Anna Cho, Junior J West, Ashleigh J. Crawford, Gabriella C. Russo, B. R. Si, Jungwoo Kim, Lauren Hoffner, Cholsoon Jang, Moonjung Jung, Robert D Leone, K. Konstantopoulos, Andrew J Ewald, Denis Wirtz, Sangmoo Jeong.(2024). E-cadherin Induces Serine Synthesis to Support Progression and Metastasis of Breast Cancer..
[29] I. O. Osman, Clémence Garrec, Gabriel Augusto Pires de Souza, A. Zarubica, Djamal Brahim Belhaouari, J. Baudoin, H. Lepidi, J. Mege, B. Malissen, B. Scola, C. Devaux.(2022). Control of CDH1/E-Cadherin Gene Expression and Release of a Soluble Form of E-Cadherin in SARS-CoV-2 Infected Caco-2 Intestinal Cells: Physiopathological Consequences for the Intestinal Forms of COVID-19.
[30] L. Thorstensen, G. E. Lind, T. Løvig, C. B. Diep, G. Meling, T. Rognum, R. Lothe.(2005). Genetic and epigenetic changes of components affecting the WNT pathway in colorectal carcinomas stratified by microsatellite instability..
[31] Hong Liu, Yan Ma, Hong-wei He, Wu-li Zhao, R. Shao.(2017). SPHK1 (sphingosine kinase 1) induces epithelial-mesenchymal transition by promoting the autophagy-linked lysosomal degradation of CDH1/E-cadherin in hepatoma cells.
[32] Xiao Tan, Wenbin Chen, D. Lv, Tao-wei Yang, Kai-hui Wu, Li-bin Zou, Junqi Luo, Xu-min Zhou, Guo-chang Liu, F. Shu, X. Mao.(2020). LncRNA SNHG1 and RNA binding protein hnRNPL form a complex and coregulate CDH1 to boost the growth and metastasis of prostate cancer.
[33] Yan He, Qimin Zhang, Yuan-Xiao Chen, Yingjian Wu, Yuan Quan, Weihua Chen, Jing Yao, Peijing Zhang.(2023). ZHX2 deficiency enriches hybrid MET cells through regulating E-cadherin expression.