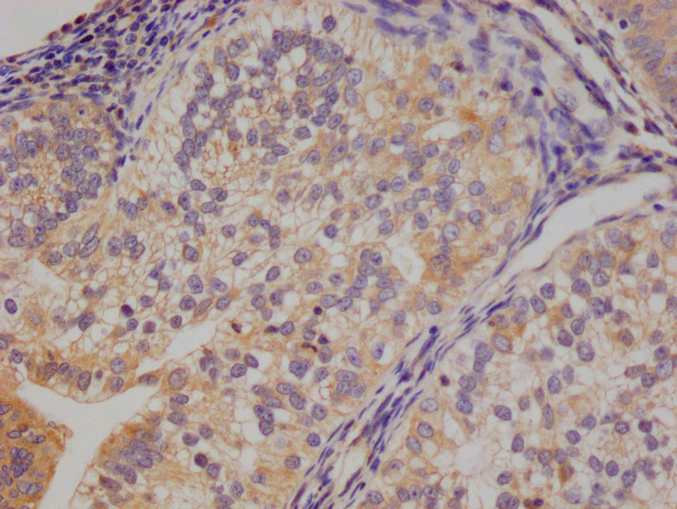
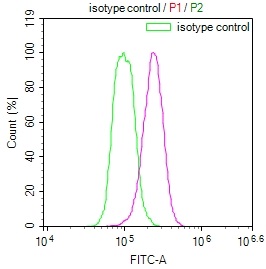
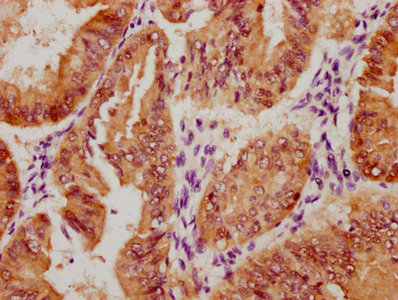
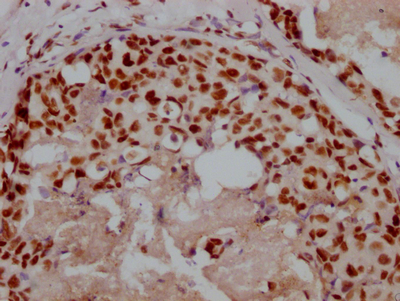
TNFSF (Sofusa anti-TNF Biosimilar) Recombinant Monoclonal Antibody
-
貨號:CSB-RA548208MB1HU
-
規格:¥83486
-
其他:
產品詳情
-
產品描述:TNFSF(Sofusa anti-TNF Biosimilar Antibody)重組單克隆抗體是一種專為科研用途開發的生物制劑。該產品基于先進的重組DNA技術制備,通過特異性靶向腫瘤壞死因子(TNF)發揮生物學作用。作為一款重組單克隆抗體,其具有高度均一的分子結構和明確的抗原結合特性,為炎癥相關疾病的機制研究、藥物靶點驗證以及體外藥效評價等科研工作提供可靠工具。 該抗體通過精準識別TNF分子的特定表位,可在體外實驗中高效阻斷TNF與其受體的相互作用,從而為探討TNF信號通路在免疫調節、炎癥反應及相關疾病發生發展中的作用機制提供有力支持。產品嚴格按照科研級標準生產,經過多輪純化和質量檢測,確保批次間的穩定性和一致性,適用于細胞實驗、蛋白結合分析、 ELISA檢測等多種實驗場景。 作為研究工具,TNFSF重組單克隆抗體有助于科研人員深入探索TNF介導的生物學過程,為開發新型抗炎藥物或治療策略提供前期研究基礎。其高度特異性和生物活性使其成為免疫學、分子生物學及藥理學等領域研究的理想選擇,支持從基礎機制研究到藥物候選分子篩選的多樣化科研需求。
-
Uniprot No.:
-
基因名:TNFSF
-
別名:Tumour necrosis factor receptor antagonist(Sorrento Therapeutics) research-grade biosimilar
-
反應種屬:Human
-
免疫原:Recombinant Human TNFSF protein
-
免疫原種屬:Homo sapiens (Human)
-
標記方式:Non-conjugated
-
克隆類型:Monoclonal
-
濃度:It differs from different batches. Please contact us to confirm it.
-
保存緩沖液:0.01M PBS,pH7.4
-
產品提供形式:Liquid
-
應用說明:Validation Status
Application-specific performance (e.g., in flow cytometry, ELISA, IHC or other assay formats) has not yet been experimentally verified by CUSABIO. Users are advised to determine the optimal working conditions empirically in their own assay systems.
Guaranteed Quality
① Antibody purity?> 95% tested by SDS-PAGE.
② Endotoxin level < 0.1EU/ug tested by LAL method. -
儲存條件:Upon receipt, store at -20°C or -80°C. Avoid repeated freeze.
-
貨期:3-4 weeks
-
用途:It is a non-therapeutic biosimilar antibody, owning the same variable region from the corresponding approved therapeutic antibody. In conclusion, it is a research-grade biosimilar antibody and expressed in mammalian cell, which can be directly used as positive controls in drug discovery or used for rapid verification of the biological functions of target protein.
Most popular with customers
-
-
YWHAB Recombinant Monoclonal Antibody
Applications: ELISA, WB, IHC, IF, FC
Species Reactivity: Human, Mouse, Rat
-
Phospho-YAP1 (S127) Recombinant Monoclonal Antibody
Applications: ELISA, WB, IHC
Species Reactivity: Human
-
-
-
-
-